- Paper Title: On the Abundances of Noble and Biologically Relevant Gases in Antarctica
- Authors: O. Mousis et al
- First Author’s Affiliation: Universite de Franche-Comte, Bensancon-Cedex, France
- Journal: Astrobiology (Accepted)
Background
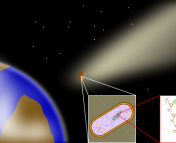
Lake Vostok is an underground (well – underice) lake in Antarctica. It’s been buried under 4 kilometers of ice for 1-15 million years, and is the closest thing we have to a real-life “Lost World”. Life in this lake, if any, has evolved in isolation for millions of years, deprived of access to sunlight and direct contact with the atmosphere. What new cellular mechanisms might have evolved here? How does isolation affect ecosystems? Questions like these are why biologists are excited about Lake Vostok – and why care is being taken to avoid contamination when drilling down to study it.

Lake Vostok, with Russian base shown. Vostok is comparable in size to Lake Ontario. Image via. NSIDC.
Astrobiologists are particularly excited about this lake because conditions in Vostok are similar to what we might expect to find on Jupiters’ moon Europa, which is thought to similarly host an oxidizing liquid water ocean protected by a layer of ice. The pressure at a depth of 30 km on Europa is equivalent to the pressure in lake Vostok. Understanding life in lake Vostok will give clues as to what kind of life might persist in Europa’s oceans.
This paper
The lake is not completely isolated from its environment. The ice above the lake is filled with bubbles of trapped air, and ice layers move down over time transporting this air. Melting at the lake/ice interface can supply dissolved gas to the lake. However, this picture is complicated by the existence of clathrates. Clathrates are crystalline structures that can trap other molecules. Depending on which atmospheric molecules are locked away inside clathrates and which are released to the lake, the abundances of dissolved gases may vary from the atmospheric abundances. To account for this effect, the team used a complex thermodynamic model for clathrate uptakes. They assumed gas-rich input from the top of the lake and gas-poor water output from the bottom of the lake, with the lake otherwise a closed system. Their work was more sophisticated relative to past work: it accounted for the effects of having multiple species of gas present in the system, and contained a more detailed and appropriate thermodynamic model.
Running their models, the authors found Xe, Kr, Ar, and CH4 to be depleted in lake water relative to atmospheric abundance due to clathrate trapping. On the other hand, the authors found an excess of CO2 relative to atmospheric abundance. These enhancements were present at the 1-2 order of magnitude level – highly significant! It’s worth noting that the values given in the paper are defined relative to present atmospheric composition, which may not be representative of the atmosphere at all times over the lake’s history. In particular, anthropogenic carbon inputs and the carbon cycle mean that the CO2 fraction might have varied by 10-30%. However, the authors argue that the conclusions of the paper with respect to the efficiency of CO2 trapping are unaffected by this: if there was more CO2 in the past, more of it should have been captured meaning the relative abundances should not have changed.
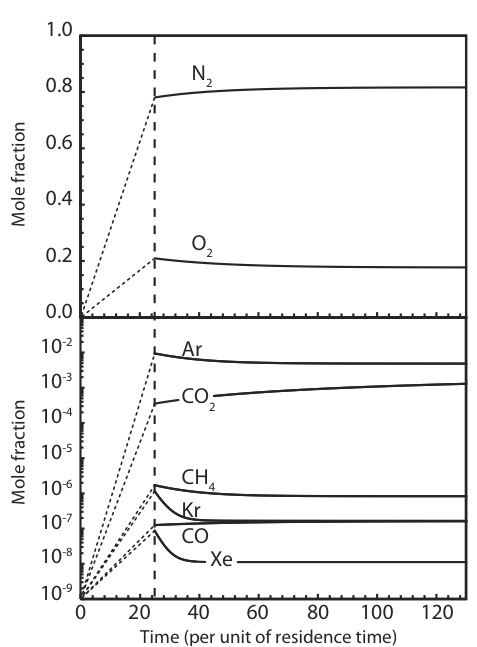
Mole fractions of different gases in Lake Vostok water over time for Type II clathrates. The results for Type I clathrates are similar. Anomalies from the initial gas abundances emerge over time.
These findings have strong implications for future studies of the lake. Deviations from these values will imply the existence of other mechanisms not accounted for in this study. For example, assuming minimal rock-water interactions, an excess of carbon dioxide and deficit of methane can be attributed to the action of methanogens which convert CH4 to CO2. Studies of the chemical properties of the lake can be used to test for life! More generally, deviations from these values can also inform our understanding of volatile transport to the lake, rock-water chemistry, and much more.