Title: The Case of H2C3O Isomers, Revisited: Solving the Mystery of the Missing Propadienone
Authors: Christopher N. Shingledecker, Sonia Álvarez-Barcia, Viktoria H. Korn, and Johannes Kästner
First Author’s Institution: Center for Astrophysics Studies Max Plank Institute for Extraterrestrial Physics & Institute for Theoretical Chemistry at the University of Stuttgart
Status: Published in the Astrophysical Journal [Closed Access], Posted on ArXiv [Open Access]
Finding and Making Molecules
Looking for different chemicals in space is a lot like searching for Waldo in the infamous search and find series “Where’s Wally?” Only imagine that the search and find page is light years away from you and all you have is a flashlight.

As our knowledge and understanding of chemical evolution in space grows, astronomers are seeking the detection of more and more complex organic molecules (COMs). Molecules that may lead to the production of life such as prebiotic molecules that may eventually form DNA, and other larger COMs are rather difficult to detect, so oftentimes we use theoretical calculations to predict the evolution and abundance of these larger molecules.
Chemical models commonly use kinetics, how energy changes over as a reaction progresses, to determine the rate at which chemical reactions occur, and thus the rate at which more complex molecules form and how abundances vary over time. Kinetics tells us that chemical reactions typically have an energy barrier to get from reactants to products. However, space is so cold that there isn’t enough energy available to overcome energy barriers (imagine pushing a 500 pound boulder over the top of Mount Everest). So, we assume that only barrier-less reactions can occur in space. There is a noteworthy exception of ultra hot regions like HII regions, supernovae and such, where temperatures are high enough to overcome reaction barriers.
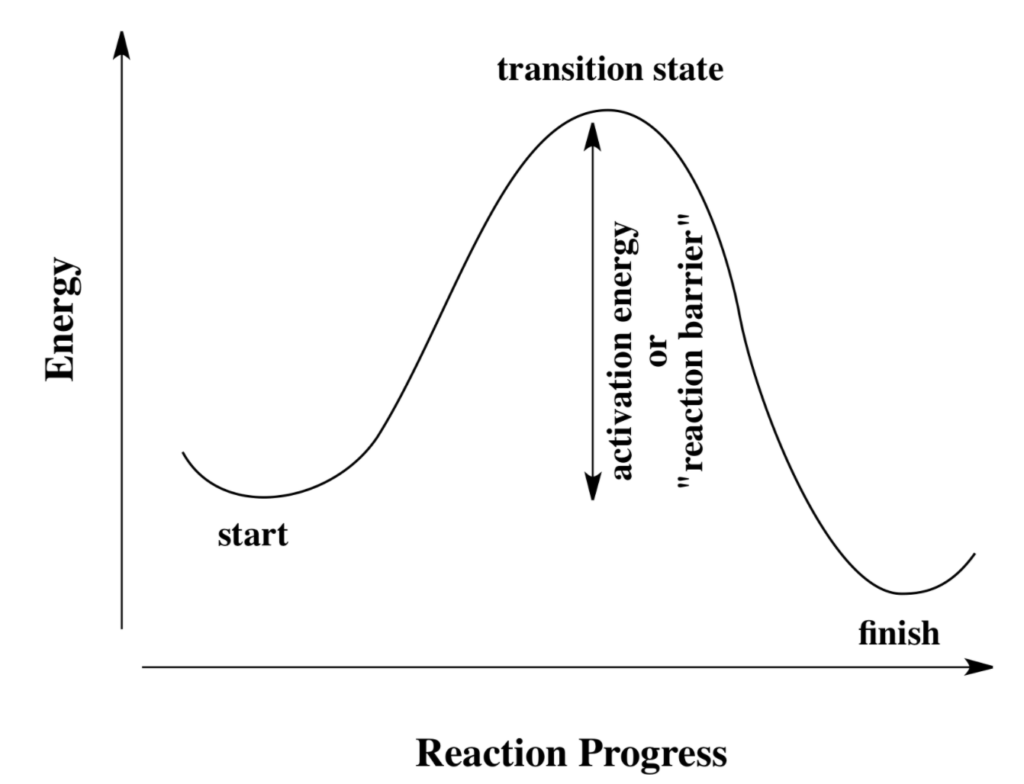
One of the most important aspects of theoretical research is matching observational data. If theoretical models using activation barriers and chemical kinetics are not able to match observations, then that usually indicates that there is a physical or chemical process that we don’t know about.
The Missing Molecule
In the last decade, one important molecule that has alluded astronomers is CH2CCO, or propadienone. CH2CCO is actually one of three different molecules that can be made from two hydrogen atoms, three carbon atoms, and one oxygen atom (H2C3O). These are known as structural isomers, meaning they’re made up of all the same atoms, but the atoms can be arranged differently to make different molecules.
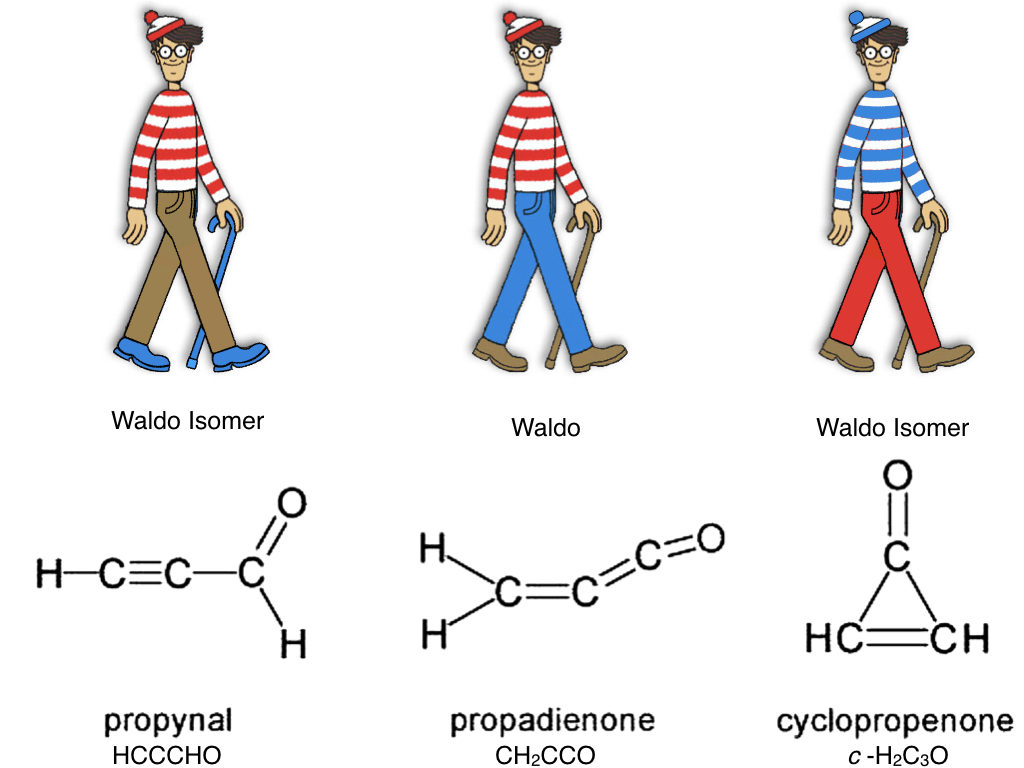
H2C3O isomer structures were found in this paper, and “Waldo” is from this webpage.
Propadienone (CH2CCO) is the most stable isomer of H2C3O, meaning CH2CCO has the lowest ground state energy and the H2C3O atoms are “happiest” in the CH2CCO configuration. According the the minimum energy principle, which uses thermodynamics rather than kinetics to predict chemical evolution, CH2CCO should be the most abundant of the three isomers, since it is the most stable of the three. Despite observational efforts and archival data searches, no one has been able to detect CH2CCO in space even though the other two H2C3O isomers have been detected. Therefore, the minimum energy principle states that CH2CCO should be detectable as well. This disagreement between observations and theory challenged the minimum energy principle and questioned the validity of relying on kinetics for chemical models.
Where’s Waldo CH2CCO?
So, where is CH2CCO? As it turns out, we still haven’t detected it in space. However, today’s paper uses theoretical calculations to find “where” CH2CCO is hiding. The authors map reactions associated with the H2C3O isomers using density functional theory (DFT). DFT uses quantum mechanics and kinetics to determine the most stable structures of molecules and their associated energies. CH2CCO can react with two hydrogen atoms to form propenal (CH2CHCHO). The process of adding a single H atom, or a proton, is a common reaction known as hydrogen addition. CH2CCO undergoes two hydrogen additions to form CH2CHCHO, both of which were found to be barrier-less reactions.
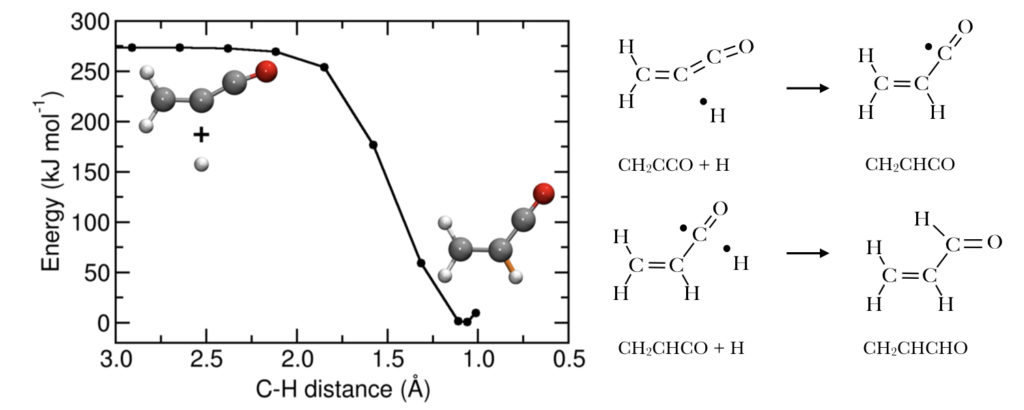
Left: Hydrogen additions to CH2CCO to form CH2CHCHO. Each reaction adds a single H atom to the carbon chain. Note the black dots are single, unpaired electrons (radicals).
Interestingly enough, hydrogen addition to the second most stable H2C3O isomer, propynal (HCCCHO), is found to have a reaction barrier. Thus propynal is able to persist in molecular clouds, while CH2CCO is converted to CH2CHCHO. These findings are consistent with both previous experimentation and observations of the Sagittarius B2 molecular cloud, where the two less stable H2C3O isomers and CH2CHCHO were detected, but CH2CCO was not.
Today’s paper shows that the “missing” molecule propadienone (CH2CCO) was never actually missing; it was just masquerading as CH2CHCHO. This discovery is important, since it shows us that kinetic theory and observations of CH2CCO are actually in agreement, rather than disagreement. Additionally, today’s paper confirms the validity of using chemical kinetics and reaction barriers (or lack of barriers) to predict chemical evolution in astronomical settings.
Sometimes search and finds, like finding molecules in astronomical settings, can be difficult, but ultimately, finding the missing pieces helps us better understand our Universe.
Now that we’ve found CH2CCO, did you find Waldo in the first figure?