Title: First Detection of the Simplest Organic Acid in a Protoplanetary Disk
Authors: Cécile Favre, Davide Fedele, Dmitry Semenov, Sergey Parfenov, Claudio Codella, Cecilia Ceccarelli, Edwin A. Bergin, Edwige Chapillon, Leonardo Testi, Franck Hersant, Bertrand Lefloch, Francesco Fontani, Geoffrey A. Blake, L. Ilsedore Cleeves, Chunhua Qi, Kamber R. Schwarz, Vianney Taquet
First Author’s Institution: National Institute for Astrophysics, Arcetri Astrophysical Observatory, Florence, Italy
Status: Published in The Astrophysical Journal Letters [open access on arXiv]
Molecules in Space
Somewhat surprisingly, space is anything but empty from a chemical point of view. Astronomers have discovered more than 200 different molecules in space – around stars, in comets, throughout molecular clouds, and even in nearby galaxies. One of the most intriguing places where molecules have been detected is in the cold gas around protoplanetary disks. Protoplanetary disks, which are composed of dense gas and dust rotating around a newly-formed star, are the sites of planet formation. Understanding the chemistry occurring in these disks is especially interesting since this chemistry influences the potential for life on resulting planets. While comparatively fewer types of molecules (now about 20) are known to exist in disks, the high sensitivity of the Atacama Large Millimeter/submillimeter Array (ALMA) is making new discoveries at an increasing rate. In today’s astrobite, we take a look at the first detection of an organic acid in a protoplanetary disk and the implications for the origins of life outside of our solar system.
Discovering Formic Acid in TW Hydrae
Today’s authors used ALMA to observe the protoplanetary disk around TW Hydrae, which at about 200 light years away is the closest known disk to Earth. TW Hydrae has been featured in several previous posts, as it has prominent rings and gaps in its dust distribution, potentially indicating on-going planet formation since newly-formed planets will sweep up and accrete dust particles. Over the last decade, TW Hydrae has also been a boon for new molecular detections around disks with ammonia (NH3), methanol (CH3OH), and water (H2O) all being first identified in this disk.
The reported discovery of trans-formic acid, or t-HCOOH, in TW Hydrae represents the first organic acid found around a young star. Moreover, formic acid is the first organic molecule containing two oxygen atoms detected in a protoplanetary disk, which shows that organic chemistry is active but difficult to detect. Importantly, t-HCOOH contains a critical chemical component that is also found in amino acids, the structural units of proteins. In fact, t-HCOOH is involved in a chemical route that produces glycine, the simplest amino acid.
How to Detect Molecules in Protoplanetary Disks?
The thermal radiation coming from the nearby, central star in a protoplanetary disk causes the surrounding gas to be hotter and have more energy than other colder or emptier regions of space. Hence, the molecules comprising this gas will be moving around and rotating due to this extra energy. These rotating molecules will occasionally spin down from a faster, higher-energy rotation state to a slower, lower-energy state. In doing so, they will emit a photon of light at a precise frequency, often in the radio or microwave region of the electromagnetic spectrum. These photons can be captured and recorded as spectral lines by radio telescopes like ALMA. Astronomers can then use this spectral information to identify and characterize these molecules. Using this technique, today’s authors were able to discover formic acid and learn how much t-HCOOH is present in TW Hydrae. Figure 1 shows a schematic of a rotational transition of formic acid and its associated spectrum shown in the paper.
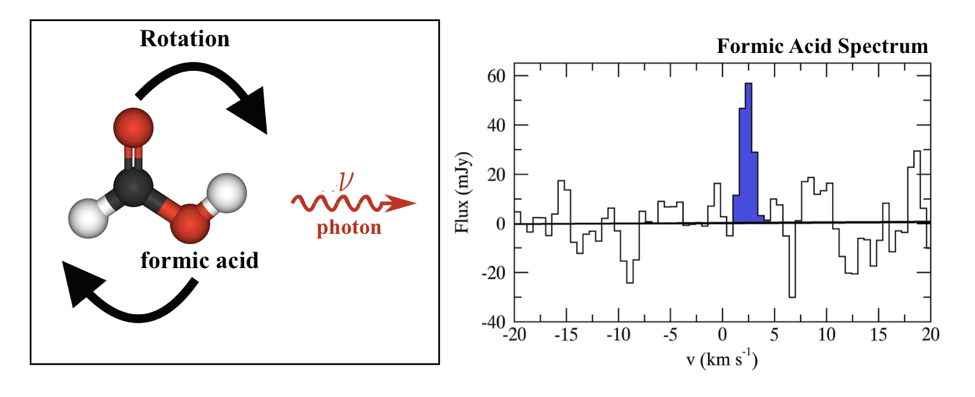
Figure 1: Left: A schematic showing the rotation of formic acid from a higher- to a lower-energy state and the subsequent emission of a photon with a specific frequency . Right: The spectrum of a rotational transition of formic acid with the spectral line colored in dark blue. The x-axis has been converted from frequency to velocity and the y-axis is given in milli Janskys, or mJy, which is a specialized unit of radio astronomy and measures how much light is received at a particular frequency. Adaptation of Figure 2 in the paper.
Observations of Formic Acid in TW Hydrae
From the properties of this spectral line, we can also infer that the emitting gas has a temperature of only 20-40 K, a reminder of the remarkably different physical conditions of these disks compared to everyday life here on Earth. The spatial distribution of formic acid is peaked toward the center of TW Hydrae but extends out beyond 200 AU, which is nearly four times the size of our solar system. A face-on view of the t-HCOOH gas in TW Hydrae is shown Figure 2. This large spatial extent is not unexpected – protoplanetary disks are often several times larger than a more evolved planetary system like our solar system. As the disks evolve, the gas and dust will be absorbed into forming planets or dispersed out into interstellar space, leaving behind a more compact planetary system.
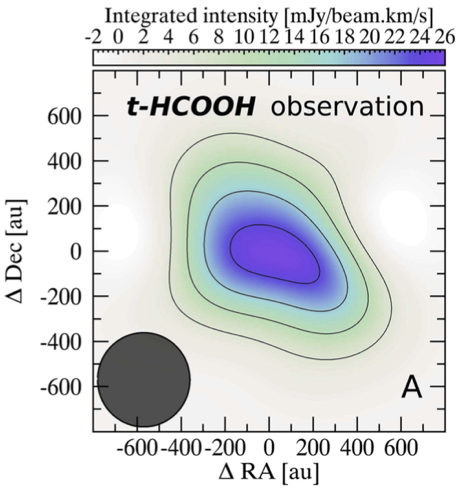
Figure 2: Face-on view of the total radio emission from t-HCOOH around the TW Hydrae disk taken with ALMA. The image is centered on the central young star in TW Hydrae. Darker colors and additional contours represent increasing strength of the signal from formic acid. The x- and y-axes are in equatorial coordinates and show a range of over 1200 AU, which corresponds to nearly 24 times the size of our solar system. The dark gray circle in the lower left indicates the resolution of the radio observation. Adaptation of Figure 3 in the paper.
The Building Blocks of Life in Space
As noted previously, molecules in the gaseous material around protoplanetary disks will eventually end up being incorporated into planets. This is why it’s especially intriguing that we now know that some of the basic building blocks of life are already present in a protoplanetary disk. It’s an exciting time to be studying the chemistry of planet formation! Not only are we finding more and more evidence for the potential for the origins of life outside of our solar system, but the detection of increasingly complex molecules like formic acid also hints at an even more complex, but currently undetected, organic chemistry going on in these disks.